Peptide researchers face a critical challenge: ensuring safety amid variable peptide quality and contamination risks. Without a structured approach to risk identification, researchers risk compromised experiments, regulatory violations, and potential clinical trial failures. This guide walks you through evidence-based methods for identifying peptide risks, from initial preparation through testing, storage protocols, and regulatory compliance, helping you confidently mitigate safety concerns.
Table of Contents
- Prerequisites And Preparations
- Methods For Peptide Risk Identification
- Storage And Handling Protocols
- Safety Testing And Risk Mitigation
- Regulatory Considerations And Compliance
- Common Mistakes And Troubleshooting
- Expected Results And Outcomes
- Explore High-Quality Peptides For Safer Research
- Frequently Asked Questions
Key takeaways
| Point | Details |
|---|---|
| Structured protocols improve safety | Validated analytical methods and systematic testing reduce peptide-related adverse events by over 50%. |
| HPLC detects impurities effectively | High-performance liquid chromatography identifies contamination at levels as low as 0.1%, ensuring purity. |
| Proper storage prevents degradation | Maintaining peptides at -20°C or below extends shelf life by approximately 6 months and preserves activity. |
| Endotoxin testing is essential | Routine pyrogenic screening prevents clinical trial halts and life-threatening adverse reactions. |
| EMA compliance ensures readiness | Following European Medicines Agency guidelines prepares research for regulatory audits and approvals. |
Prerequisites and preparations
Before diving into peptide risk identification, you need the right foundation. Having access to validated peptide sequence databases ensures you can verify peptide identity and known risk profiles accurately. Consulting validated databases like the European Pharmacopoeia helps reduce errors in peptide selection and cross-reference safety information.
You also need basic training in analytical methods. Proficiency with high-performance liquid chromatography and mass spectrometry is non-negotiable for detecting impurities and modifications. If your facility lacks in-house equipment, establish partnerships with certified testing laboratories that can perform these analyses reliably.
Endotoxin testing capabilities are another critical requirement. Whether through contracted services or in-house kits, you must be able to screen for pyrogenic contaminants before any clinical application. Familiarity with EMA regulatory guidelines ensures you understand compliance thresholds and documentation standards.
Key preparation steps include:
- Securing access to validated peptide databases for sequence verification
- Obtaining training in HPLC and mass spectrometry techniques
- Establishing endotoxin testing capabilities through labs or kits
- Reviewing current EMA guidelines on peptide purity and safety
- Identifying certified testing laboratories for outsourced analysis
Pro Tip: Create a pre-assessment checklist covering equipment access, database subscriptions, and staff training status. This saves time when starting new peptide projects and prevents overlooked preparation gaps.
Methods for peptide risk identification
Accurate risk identification relies on robust analytical methods. High-performance liquid chromatography is the gold standard for assessing peptide purity, capable of detecting impurities at concentrations as low as 0.1%. HPLC separates peptide components based on their chemical properties, revealing synthesis by-products, degradation fragments, and contaminants that compromise safety.
Mass spectrometry complements HPLC by providing molecular weight data and identifying specific modifications or degradation products. This technique confirms peptide identity and detects post-translational modifications that may alter biological activity. Together, these methods form a comprehensive analytical strategy.
Endotoxin testing screens for bacterial lipopolysaccharides that trigger dangerous pyrogenic responses. Visual and physical inspection serves as a preliminary step, identifying obvious discoloration, precipitation, or particulate matter before proceeding to detailed analysis. These healing peptide research techniques apply across various peptide classes.
| Method | Detection Limit | Primary Use | Speed |
|---|---|---|---|
| HPLC | 0.1% impurities | Quantitative purity analysis | 30-60 min |
| Mass Spectrometry | Single modifications | Molecular weight verification | 15-30 min |
| Endotoxin Testing | 0.01 EU/mL | Pyrogenic contamination | 30-45 min |
| Visual Inspection | Visible particles | Preliminary screening | 2-5 min |
Core analytical methods include:
- Running HPLC to quantify impurity levels and verify purity thresholds
- Performing mass spectrometry to confirm molecular weight and detect modifications
- Conducting endotoxin testing to screen for pyrogenic contaminants
- Starting with visual inspection to catch obvious quality issues early
- Cross-referencing results with manufacturer certificates of analysis
Pro Tip: Always run a reference standard alongside your test sample. This internal control validates your analytical system performance and catches instrument drift before it compromises your risk assessment.
Storage and handling protocols
Maintaining peptide quality demands strict storage conditions. Improper storage causes peptides stored above -20°C to show up to 30% loss in activity within one month, dramatically increasing risk profiles. Freezers set to -20°C or below minimize degradation and preserve peptide integrity over extended periods.

Using desiccants and inert gas atmospheres provides additional protection. Moisture accelerates hydrolytic degradation, while oxygen promotes oxidation of sensitive amino acids like methionine and cysteine. Vacuum-sealing peptides with desiccant packets and storing under nitrogen or argon atmospheres extends stability significantly.
Regular monitoring prevents silent degradation. Schedule monthly checks of freezer temperatures, inspect vial seals for integrity, and maintain detailed logs documenting storage conditions. This peptide storage documentation creates an audit trail and enables quick identification of storage failures.
Optimal storage practices include:
- Maintaining consistent storage temperatures at -20°C or colder
- Using desiccants and inert gas atmospheres to prevent moisture and oxidation
- Implementing monthly temperature monitoring and documentation
- Avoiding freeze-thaw cycles by aliquoting peptides into single-use portions
- Labeling vials with receipt dates and first-use dates for rotation
Pro Tip: Divide bulk peptide stocks into small aliquots immediately upon receipt. Single-use aliquots eliminate repeated freeze-thaw cycles that cause cumulative degradation, even when stored at correct temperatures.
Safety testing and risk mitigation
Comprehensive safety testing starts with endotoxin screening. Failure to perform endotoxin testing before clinical use has resulted in clinical trial halts due to pyrogenic adverse events. EMA guidelines set limits at 5 endotoxin units per kilogram of body weight, and your testing must verify compliance before any human application.
Supplier validation prevents contamination at the source. Request certificates of analysis showing purity testing, endotoxin levels, and analytical method validation. Audit supplier quality management systems and verify their adherence to Good Manufacturing Practices. This peptide safety testing approach reduces downstream safety incidents.
Systematic checklists track safety steps and prevent oversights. Document every test performed, record results against acceptance criteria, and maintain chain of custody records for regulatory audits. Monitor safety throughout handling and experimental procedures, watching for unexpected reactions or stability issues.
Essential safety measures include:
- Performing routine endotoxin testing meeting EMA limits of 5 EU/kg
- Validating suppliers through certificates and quality system audits
- Using detailed checklists to track all safety testing steps
- Monitoring peptide stability during storage and handling
- Documenting all safety tests with dated records and signatures
Safety testing workflow:
- Receive peptide and verify supplier documentation immediately
- Perform visual inspection for particulates or discoloration
- Conduct HPLC analysis to confirm purity specifications
- Run endotoxin testing and verify results meet regulatory limits
- Document all findings and approve for use or reject as needed
Warning: Skipping endotoxin testing to save time or cost creates catastrophic risk. Pyrogenic reactions can be life-threatening and have stopped clinical programs completely. Never compromise on this critical safety step.
Regulatory considerations and compliance
European Medicines Agency guidelines establish clear standards for peptide impurities and endotoxin limits. EMA requires detailed documentation of all testing procedures, results, and corrective actions. Your records must demonstrate traceability from raw materials through final peptide application, creating a complete quality history.
Understanding the distinction between research-grade and clinical-grade peptides matters for compliance planning. Research-grade materials face fewer documentation requirements but still demand purity verification. Clinical-grade peptides require full regulatory documentation, validated manufacturing processes, and extensive safety testing. Plan your compliance approach based on intended use.
Compliance timelines vary by application. Research use may require weeks for basic documentation, while clinical applications need months of preparation including manufacturing validation, stability studies, and regulatory submissions. Preparing for audits means organizing records systematically, training staff on documentation standards, and conducting internal audits to identify gaps. The retatrutide mechanism and compliance context illustrates these regulatory considerations.
Regulatory compliance essentials include:
- Following EMA guidelines on maximum impurity levels and endotoxin thresholds
- Maintaining complete documentation of all testing and handling procedures
- Understanding research-grade versus clinical-grade regulatory requirements
- Planning compliance timelines based on intended peptide applications
- Conducting regular internal audits to maintain regulatory readiness
Steps to maintain regulatory readiness:
- Review current EMA guidance documents annually for updates
- Implement standardized documentation templates across all projects
- Train all staff on proper record-keeping and compliance procedures
- Schedule quarterly internal audits of documentation and processes
- Establish relationships with regulatory consultants for expert guidance
Common mistakes and troubleshooting
Skipping supplier validation ranks among the most dangerous errors. A survey found that 40% of peptide-related adverse events stemmed from contamination with synthesis by-products, highlighting critical importance of vendor selection. Always verify supplier credentials, request third-party testing, and audit quality systems before committing to a peptide source.
Neglecting endotoxin testing stops clinical programs in their tracks. Researchers focusing solely on purity sometimes overlook pyrogenic screening, only to face trial suspensions when safety monitoring detects problems. Make endotoxin testing a mandatory checkpoint that cannot be bypassed under time pressure.
Improper storage causes silent degradation that compromises experiments. Storing peptides in standard freezers without temperature monitoring allows fluctuations that accelerate breakdown. Incomplete documentation creates compliance nightmares during audits, forcing researchers to repeat work or discard batches entirely. These wolverine stack peptide research examples demonstrate common pitfalls and solutions.
Frequent errors to avoid:
- Purchasing from unvalidated suppliers without quality documentation
- Skipping endotoxin testing due to time constraints or cost concerns
- Storing peptides in non-dedicated freezers with poor temperature control
- Failing to document testing results and storage conditions systematically
- Ignoring early warning signs like discoloration or precipitation
Troubleshooting contamination:
- Request replacement batches with full certificates of analysis from suppliers
- Perform independent third-party testing to verify supplier claims
- Review handling procedures for potential contamination sources
- Implement additional filtration steps if bacterial contamination suspected
- Document all contamination incidents and corrective actions taken
Expected results and outcomes
Comprehensive peptide risk identification protocols require 2-4 weeks for full validation, including testing, documentation, and regulatory compliance checks. This timeline allows thorough analytical work without rushing critical safety steps. Expect initial setup to take longer as you establish procedures, but subsequent assessments become faster as systems mature.
Implementing rigorous protocols yields measurable safety improvements. Facilities following structured risk identification see more than 50% reduction in peptide-related adverse events compared to those using ad hoc approaches. Proper storage extends peptide shelf life by approximately 6 months, reducing waste and improving research economics.
Regulatory compliance success rates improve dramatically with systematic risk management. Programs with documented risk identification achieve smoother audits, faster regulatory approvals, and fewer compliance violations. These retatrutide research outcomes demonstrate measurable benefits of rigorous safety protocols.
| Metric | Target Threshold | Measurement Method |
|---|---|---|
| Peptide Purity | ≥98% by HPLC | Chromatographic peak integration |
| Endotoxin Level | <5 EU/kg | LAL or chromogenic assay |
| Storage Stability | <10% activity loss/6 months | Periodic bioassay testing |
| Documentation Completeness | 100% records filed | Audit checklist verification |
| Supplier Quality Score | ≥90% compliance | Quarterly performance review |
Expected outcomes include:
- Complete risk assessment within 2-4 weeks for new peptide batches
- Over 50% reduction in safety incidents with validated protocols
- Extended peptide shelf life by approximately 6 months through proper storage
- Higher regulatory approval rates for compliant research programs
- Clear audit trails enabling rapid regulatory inspection responses
Explore high-quality peptides for safer research
Now that you understand comprehensive peptide risk identification, sourcing reliable peptides becomes your next priority. CK Peptides provides research-grade peptides with purity exceeding 99%, backed by complete certificates of analysis and rigorous quality testing. Every batch undergoes HPLC verification and endotoxin screening, giving you confidence in peptide safety from the start.
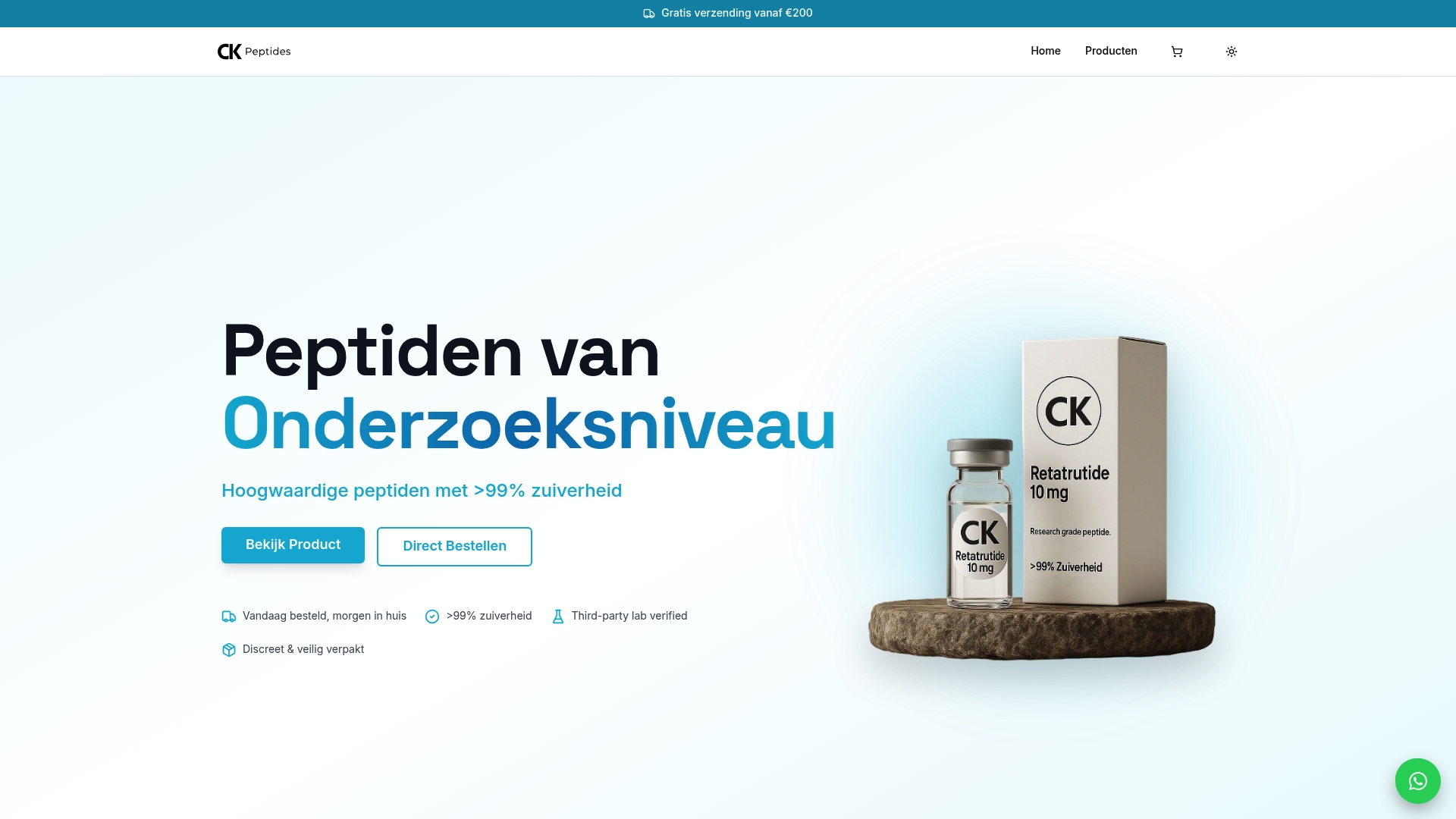
Our research starter kit includes everything needed to begin peptide experiments with proper safety protocols already in place. Products comply with European regulatory standards, reducing your compliance workload and supporting the systematic risk management approach detailed in this guide. For researchers exploring metabolic peptides, our retatrutide 10mg peptide offers pharmaceutical-grade quality ideal for advanced studies. Reliable sourcing eliminates the supplier validation challenges discussed earlier, letting you focus on research rather than risk mitigation.
Frequently asked questions
What equipment is essential for peptide risk identification?
Essential equipment includes high-performance liquid chromatography systems, mass spectrometry instruments, and endotoxin testing kits or access to contracted laboratory services. Proper infrastructure for controlled storage at -20°C or below is equally critical. Many researchers use a combination of in-house capabilities for routine testing and specialized labs for advanced analyses, balancing cost with comprehensive peptide research needs.
How does improper storage impact peptide safety?
Improper storage above -20°C causes rapid peptide degradation, with peptides losing up to 30% activity within one month. This degradation increases safety risks by creating unknown breakdown products that may trigger unexpected biological responses. Temperature fluctuations also promote aggregation and oxidation, compromising experimental reproducibility. Implementing strict peptide storage protocols prevents these quality failures and maintains peptide integrity throughout research timelines.
Why is endotoxin testing critical before clinical peptide use?
Endotoxin testing prevents life-threatening pyrogenic reactions including fever, hypotension, and systemic inflammatory responses in clinical applications. Clinical trials have been halted due to lack of endotoxin testing leading to pyrogenic adverse events, causing massive financial losses and regulatory setbacks. Even research-grade peptides used in animal studies benefit from endotoxin screening to prevent confounding inflammatory responses. Following proper peptide safety testing protocols protects both research subjects and program viability.
How long does comprehensive peptide risk identification typically take?
Complete risk assessment protocols usually take 2-4 weeks for full validation including analytical testing, documentation, and regulatory compliance verification. Initial assessments take longer as you establish procedures and baselines, while subsequent batch testing becomes more efficient. Rush timelines compromise thoroughness and increase risk of missing critical safety issues. Planning adequate time for comprehensive evaluation ensures reliable results and supports successful retatrutide peptide research and other advanced peptide studies.
Recommended
- Complete Guide to Retatrutide: Mechanism, Research, and Metabolic Peptide Science | CK Peptides Blog
- Retatrutide Peptide: The Breakthrough Triple-Hormone Research Changing Obesity Science | CK Peptides Blog
- The Wolverine Stack Explained: BPC-157 & TB-500 Peptide Research | CK Peptides Blog
- BPC-157: The Healing Peptide Explained by Science
